#fasttrackdesignation search results
📢 Revolutionary Therapy Receives FDA Fast Track! #AdrenomedAG #FDA #fasttrackdesignation #septicshocktherapy bioprocessupdates.com/companies/revo…
bioprocessupdates.com
ᐈ Revolutionary Therapy Receives FDA Fast Track! - BioprocessUpdates
🎯 Shocking news: German biotech Adrenomed's septic shock therapy gets FDA fast track designation! 🚀💊🩸 Exciting potential for effective treatment! 💡
#Evaxion receives #FDA #fasttrackdesignation for #personalizedcancer #immunotherapy $EVAX 1stoncology.com/blog/evaxion-r…
There are ~150,000 patients suffering from EGFRi rash in the U.S. @azitrainc has received #FastTrackDesignation from the #FDA for #EGFRiAssociatedRash; plans to initiate a Ph1/2 clinical study in patients by mid 2025. #EGFRirash #Dermatology #PrecisionDerm #FastTrack

📣The @US_FDA granted #FastTrackDesignation for AVB-001. Read full article & learn more about our #clinicaltrial at prnewswire.com/news-releases/…

J. Robert Coleman, CEO of Codagenix, weighs on the importance of developing an #RSV #vaccine and talks about the company’s candidate CodaVax-RSV which received #fasttrackdesignation. Read more at @Contagion_Live -> contagionlive.com/view/fda-fast-…

The #FDA granted #FastTrackDesignation for quaratusugene ozeplasmid, with atezolizumab, in patients with extensive-stage small cell #lungcancer who did not develop #tumor progression after receiving atezolizumab and #chemotherapy: ow.ly/zE7j50Pu9aF #CancerResearch
#BicycleTherapeutics Announces #FDA #FastTrackDesignation Granted to# BT8009 for the Treatment of Adult Patients with Previously Treated Locally Advanced or Metastatic #UrothelialCancer $BCYC 1stoncology.com/blog/bicycle-t…
Today, we announced that the @FDA has granted #FastTrackDesignation to our clinical program evaluating #pemvidutide, our novel #GLP1/Glucagon agonist, for the treatment of #NASH. Learn more: bit.ly/462jQQp.
#DualityBio Announces #DB1303 Granted #FastTrackDesignation by the U.S. Food and Drug Administration (#FDA) for the Treatment of Advanced, Recurrent or #Metastatic #EndometrialCarcinoma with #HER2Overexpression 1stoncology.com/blog/dualitybi…
Selinexor Receives Fast Track Designation From FDA For Myelofibrosis Treatment Read more at our website link in bio! #fasttrackdesignation #FDA #myelofibrosis #selinexor #xpovio #alzheimersdisease #cumulusneuroscience #diagnosis #digitalhealth #electroencephalogram #gadgets

#CaribouBiosciences Announces #FDA Granted #FastTrackDesignation to #CB011 an #Allogeneic #CART #CellTherapy for #Relapsed or #Refractory #MultipleMyeloma $CRBU 1stoncology.com/blog/caribou-b…
We’ve launched REO 033, a Phase 2 study of #pelareorep + bevacizumab & FOLFIRI in second line KRAS mutant, microsatellite stable #ColorectalCancer. Following FDA #FastTrackDesignation, preliminary results are expected by year end 2026. Learn more: bit.ly/4aTrzG9

#ArtivaBiotherapeutics Receives #FDA #FastTrackDesignation for #AB101 1stoncology.com/blog/artiva-bi…
.@EdgewiseTx Receives U.S. #FDA #FastTrackDesignation for EDG-5506 for the Treatment of #Duchenne Muscular #Dystrophy - check the link for more ow.ly/t1wS50QBfq6 #rarediseases and #orphandrugs

THE FDA EXPEDITES THE DEVELOPMENT OF GSK’S VACCINE FOR GONORRHEA Read more at our website link in bio! . . . #fasttrackdesignation #FDA #gonorrhea #GSK #vaccine #pharmaindustry #pharmanews #pharmaupdates #news #pharmtales

🧵 Appeals Court Upholds FDA's Denial of Fast Track Designation for Vanda's Gastroparesis Drug #Fda #VandaPharmaceuticals #FastTrackDesignation #Gastroparesis #DrugApprovalProcess
#Genexine Receives #FastTrackDesignation from Korean Health Authority (#MFDS) for its promising #DNAVaccine for advanced #CervicalCancer 1stoncology.com/blog/genexine-…
#TreadwellTherapeutics Announces #FastTrackDesignation Granted by the #FDA to #CFI402257 for the Treatment of #ERPositive/#HER2Negative #BreastCancer 1stoncology.com/blog/treadwell…
#ImmPACT Bio Granted #FDA #FastTrackDesignation for #IMPT314 in Patients with Relapsed or Refractory #AggressiveBCellLymphoma 1stoncology.com/blog/immpact-b…
We’ve launched REO 033, a Phase 2 study of #pelareorep + bevacizumab & FOLFIRI in second line KRAS mutant, microsatellite stable #ColorectalCancer. Following FDA #FastTrackDesignation, preliminary results are expected by year end 2026. Learn more: bit.ly/4aTrzG9

The FDA has granted #pelareorep #FastTrackDesignation for the treatment of 2L KRAS-mutant, microsatellite-stable metastatic #ColorectalCancer based on it achieving 2-3X the efficacy benchmarks vs. standard of care. Read more in our press release: bit.ly/4ki6lED
✅ The @US_FDA granted #fasttrackdesignation to ZEN-3694 for use alongside abemaciclib in previously treated NUT carcinoma. ✅ This combination has improved responses vs BET inhibitors alone. ✅ZEN-3694 is being evaluated in a phase 1 trial. Read more 🗞️: onclive.com/view/fda-grant…
Radiopharm Theranostics (ASX: RAD) Gains FDA Fast Track for RAD101, Shares Jump 12.5% @TeamRadiopharm #FastTrackDesignation #ASX $RAD zurl.co/BPdII
There are ~150,000 patients suffering from EGFRi rash in the U.S. @azitrainc has received #FastTrackDesignation from the #FDA for #EGFRiAssociatedRash; plans to initiate a Ph1/2 clinical study in patients by mid 2025. #EGFRirash #Dermatology #PrecisionDerm #FastTrack

🧵 Appeals Court Upholds FDA's Denial of Fast Track Designation for Vanda's Gastroparesis Drug #Fda #VandaPharmaceuticals #FastTrackDesignation #Gastroparesis #DrugApprovalProcess
.@AcceleronPharma's drug #ACE-083 receives #FastTrackDesignation for the treatment of #FSHD. Find out more here>> musculardystrophyuk.org/news/breaking-…

Selinexor Receives Fast Track Designation From FDA For Myelofibrosis Treatment Read more at our website link in bio! #fasttrackdesignation #FDA #myelofibrosis #selinexor #xpovio #alzheimersdisease #cumulusneuroscience #diagnosis #digitalhealth #electroencephalogram #gadgets

US FDA Awards Fast Track Designation To Theriva Biologic's VCN-01 News: qrcd.org/5Kkx @TOVXBiologics #ClinicalStageCompany #ClinicalDevelopment #FastTrackDesignation #FoodandDrugAdministration

@uniQure_NV Receives #FDA #FastTrackDesignation for AMT-130 #GeneTherapy for the Treatment of #HuntingtonsDisease ow.ly/JKbw30onlVU #orphandrug #raredisease

#Zika vaccine candidate receives @US_FDA #fasttrackdesignation @TakedaPharma #TAK-426 bit.ly/2GrSIj7

📣The @US_FDA granted #FastTrackDesignation for AVB-001. Read full article & learn more about our #clinicaltrial at prnewswire.com/news-releases/…

J. Robert Coleman, CEO of Codagenix, weighs on the importance of developing an #RSV #vaccine and talks about the company’s candidate CodaVax-RSV which received #fasttrackdesignation. Read more at @Contagion_Live -> contagionlive.com/view/fda-fast-…

Moleculin Biotech, Inc. received Fast Track Designation from the #FDA for the treatment of soft tissue #sarcoma (STS) lung metastases. Read more at ow.ly/UBZa50EestJ #fasttrackdesignation #regulatoryaffairs #drugdevelopment #biotech #cancer #oncology
THE FDA EXPEDITES THE DEVELOPMENT OF GSK’S VACCINE FOR GONORRHEA Read more at our website link in bio! . . . #fasttrackdesignation #FDA #gonorrhea #GSK #vaccine #pharmaindustry #pharmanews #pharmaupdates #news #pharmtales

We received #FastTrackDesignation from the U.S. #FDA for our non-genetically modified #placental derived NK #CellularTherapy in development for the treatment #AML. Learn more about the potential of our #CellularTherapy candidate here: bit.ly/3ewb8RU

Zynerba Jumps on Fast Track Designation Approval bit.ly/innd_lszynerba @ZynerbaPharma #biotech #FDA #FastTrackDesignation
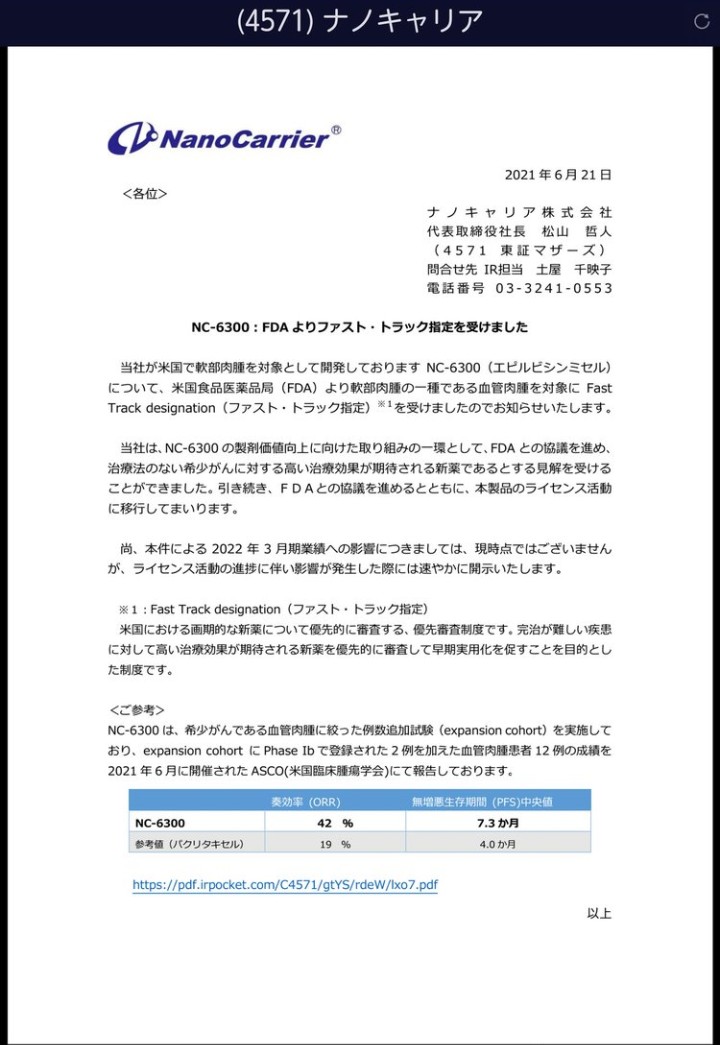
前乙です! Enjinで32円抜き出来たけどナノキャリアでJCしましたw 〈4571〉ナノキャリアの材料は↓ NC-6300: FDAよりファスト・トラック指定を受けました。 #NanoCarrier #FastTrackdesignation #FDA nanocarrier.co.jp
.@KrystalBiotech Receives #FDA #FastTrackDesignation for Inhaled #Oncology Candidate KB707 to Treat Solid #Tumors of the #Lung - check the link for more ow.ly/PqAY50QBf3R #rarediseases #orphandrugs
We’re thrilled to share that #FDA has granted #FastTrackDesignation to NUZYRA® (omadacycline) for the treatment of pulmonary Nontuberculous Mycobacterial (NTM) disease caused by Mycobacterium avium complex (MAC) & Mycobacterium abscessus (MAB). More at: bit.ly/39FSEzO

We are thrilled to announce we have received #FastTrackDesignation from the #FDA for our non-genetically modified #placenta derived NK #CellTherapy for the #treatment of adults with #GBM. Learn more: celularity.com/celularity-ann…

There are ~150,000 patients suffering from EGFRi rash in the U.S. @azitrainc has received #FastTrackDesignation from the #FDA for #EGFRiAssociatedRash; plans to initiate a Ph1/2 clinical study in patients by mid 2025. #EGFRirash #Dermatology #PrecisionDerm #FastTrack

We’re thrilled to share that the #FDA has granted #FastTrackDesignation to AD109, our investigational combination therapy that has the potential to be the first oral pharmacologic that treats #ObstructiveSleepApnea. Learn more: ow.ly/NUWa50JJsMn #OSA #sleepdisorders

We’ve launched REO 033, a Phase 2 study of #pelareorep + bevacizumab & FOLFIRI in second line KRAS mutant, microsatellite stable #ColorectalCancer. Following FDA #FastTrackDesignation, preliminary results are expected by year end 2026. Learn more: bit.ly/4aTrzG9

Something went wrong.
Something went wrong.
United States Trends
- 1. Moana N/A
- 2. Homeward Bound N/A
- 3. Election Day N/A
- 4. Maui N/A
- 5. The Rock N/A
- 6. Apple Maps N/A
- 7. #STREAMINGARIRANG N/A
- 8. #NationalPuppyDay N/A
- 9. The ACA N/A
- 10. #HealthcareNotWarfare N/A
- 11. Graceland N/A
- 12. Corgi N/A
- 13. Valerie Perrine N/A
- 14. TACO N/A
- 15. Delane N/A
- 16. Leonid Radvinsky N/A
- 17. Ty Simpson N/A
- 18. Dwayne Johnson N/A
- 19. #NoHayAmnistíaParaCriminales N/A
- 20. Reacher N/A
























