#ondls search results
India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

Since 2025, all state-level #PharmaLicenses in India—manufacturing, wholesale, retail & loan—must be filed via the #ONDLS portal. It standardizes approvals, renewals & inspections, with #Pharmacist requirements & 5-year validity. Follow for more ⤵️ ow.ly/HGj450X4LsV

India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

Drug quality: Hopes now hinge on @MoHFW_INDIA minister @mansukhmandviya three-pronged strategy: state-centre inspection squads; capacity building with the Hyd one health facility; unified portal for all stakeholders & strengthening #ONDLS @FinancialXpress financialexpress.com/healthcare/pha…
India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

Enhancing Drug Licensing Efficiency! BCDA President Parsan Kumar Singh & Admin Sec. Shri Tarun Ji attended a Health Dept. program with senior officials. The ONDLS system will simplify & ensure transparency in drug licensing, launching across Bihar from April 1st! #ondls

@CDSCO_INDIA_INFO We are a new LVP manufacturers from Maharashtra. Requesting to help us apply for fresh LVP manufacturing licence 28D on ONDLS portal. Unable to do so for the past 15 days due technical issue with ONDLS website. #cdsco #ondls #lvp
India Launches ONDLS to Curb Toxic Cough Syrup Tragedies #WordOfIndia #WOI #ONDLS #CDSCO #PharmaSafety #DrugRegulation #HealthIndia #DigitalGovernance #WHO
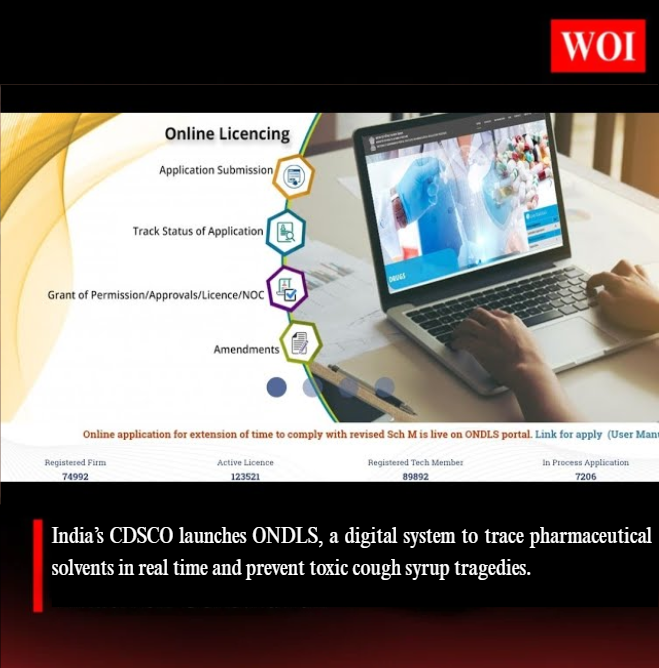
India Launches ONDLS to Curb Toxic Cough Syrup Tragedies #WordOfIndia #WOI #ONDLS #CDSCO #PharmaSafety #DrugRegulation #HealthIndia #DigitalGovernance #WHO

Since 2025, all state-level #PharmaLicenses in India—manufacturing, wholesale, retail & loan—must be filed via the #ONDLS portal. It standardizes approvals, renewals & inspections, with #Pharmacist requirements & 5-year validity. Follow for more ⤵️ ow.ly/HGj450X4LsV

India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

@CDSCO_INDIA_INFO We are a new LVP manufacturers from Maharashtra. Requesting to help us apply for fresh LVP manufacturing licence 28D on ONDLS portal. Unable to do so for the past 15 days due technical issue with ONDLS website. #cdsco #ondls #lvp
Enhancing Drug Licensing Efficiency! BCDA President Parsan Kumar Singh & Admin Sec. Shri Tarun Ji attended a Health Dept. program with senior officials. The ONDLS system will simplify & ensure transparency in drug licensing, launching across Bihar from April 1st! #ondls

Drug quality: Hopes now hinge on @MoHFW_INDIA minister @mansukhmandviya three-pronged strategy: state-centre inspection squads; capacity building with the Hyd one health facility; unified portal for all stakeholders & strengthening #ONDLS @FinancialXpress financialexpress.com/healthcare/pha…
India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

Since 2025, all state-level #PharmaLicenses in India—manufacturing, wholesale, retail & loan—must be filed via the #ONDLS portal. It standardizes approvals, renewals & inspections, with #Pharmacist requirements & 5-year validity. Follow for more ⤵️ ow.ly/HGj450X4LsV

India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

Enhancing Drug Licensing Efficiency! BCDA President Parsan Kumar Singh & Admin Sec. Shri Tarun Ji attended a Health Dept. program with senior officials. The ONDLS system will simplify & ensure transparency in drug licensing, launching across Bihar from April 1st! #ondls

India Launches ONDLS to Curb Toxic Cough Syrup Tragedies #WordOfIndia #WOI #ONDLS #CDSCO #PharmaSafety #DrugRegulation #HealthIndia #DigitalGovernance #WHO

India has strengthened #PharmaCompliance with upgrades to the #SUGAMPortal and mandatory #ONDLS platform, streamlining approvals, licensing, and clinical trial processes. Firms must adapt to dual systems for faster, predictable compliance. Learn more ⤵️ ow.ly/TZ5450WWxsi

Something went wrong.
Something went wrong.
United States Trends
- 1. #LOVEISBLINDreunion N/A
- 2. Kawhi N/A
- 3. Omar López N/A
- 4. #Survivor50 N/A
- 5. #AEWDynamite N/A
- 6. Eugenio N/A
- 7. Salvador Pérez N/A
- 8. Jordan Clarkson N/A
- 9. Massie N/A
- 10. Brittany N/A
- 11. Devonta N/A
- 12. Jordan Miller N/A
- 13. Jake Paul N/A
- 14. Italy N/A
- 15. Djokovic N/A
- 16. Vinnie Pasquantino N/A
- 17. Priyanka N/A
- 18. Japón N/A
- 19. Amber N/A
- 20. #FriendsFromYourHometown N/A








