#21cfrpart11 search results
📣 We are live! Join us now in our live showcase to learn how to ensure #DataIntegrity of Printed Documents with Scilife. Access directly here ➡️ bit.ly/prshowcase #21CFRPart11 #Annex11

Our lab solutions are like having ten smart lab assistants, except they never take a day off. #labautomation #dataintegrity #21cfrpart11 #laboratory #software

Is your lab audit-ready? 21 CFR Part 11 is more than a checkbox—it’s your foundation for trusted electronic records & signatures. Crucial for pharma, biotech, & diagnostics labs. lablynx.com/resources/arti… #21CFRPart11 #LabCompliance #LIMS #LifeSciences #LabManagement
lablynx.com
Ensure 21 CFR Part 11 Compliance with Lab Software Solutions
Meet FDA regulations with secure laboratory software designed for 21 CFR Part 11 compliance. Ensure data integrity, audit trails, and electronic signatures.
Validation of Excel-Based Solutions: Click the link below to read more: linkedin.com/posts/hemantch… #Genbioca #ExcelValidation #21CFRPart11 #DataProtectionSettings
21 CFR Part 11: more than just compliance, it’s your audit armor. Implement or upgrade your environmental monitoring with Rees EMS to fully align with 21 CFR Part 11 requirements. Read the full blog here: hubs.li/Q03tzj7z0 #rees #ems #21CFRPart11

The Power Of A No Code Platform & Solutions, It Really Matters! cloudtheapp.com/power-of-a-no-… #21CFRPart11 #Cloudtheapp #Compliance #Configurable #NoCode #QMS #QualityManagementSystem #Validation

We are excited to announce our collaboration with #Pharmagraph which offers complete #21CFRpart11 compliant solutions for clean room and facility monitoring systems. This #integration brings us closer to establishing a collaborative ecosystem with key #CGT providers.
#Celltherapy manufacturing & #21CFRPart11 compliance: Overview & considerations for instrument software In this #webinar, @thermofisher will share insights to help you better understand how electronic records & 21 CFR Part 11 compliance are related bit.ly/3lzrH6y

New case study! 🚀 See how we transformed a legacy instrument software into a 21 CFR Part 11-ready solution with secure user management, audit trails & e-signatures, without hurting usability. hubs.ly/Q03TYWSh0 #BioSistemika #LabSoftware #21CFRPart11

Pharma signing demands intent, accountability, and full traceability—not just a digital signature. CryptoESIGN CFR ensures every approval is compliant, auditable, and regulation-ready from start to finish. cryptoesign.com #CryptoSIGN #21CFRPart11 #digitalSignatures

Learn why the 21 CFR Part 11 regulation can benefit you, even if your #manufacturing enterprise is not in the medical device or pharmaceutical industries. The best practices can have universal benefits: hubs.li/Q01JLdCp0 #automation #21CFRpart11 #Ignition @InductiveAuto
I've been drawn back into detailed Part 11 discussions recently... what's old is new again (to some anyway)! #Part11 #21cfrPart11 ...and still as relevant as the predicate rules. #GMP #21cfrPart210 #21cfrPart211
Kaleido™ software’s intuitive interface makes setting up the EnVision® Nexus™ #platereader seamless. MyAssays® Desktop Pro analysis software integrates w/Kaleido, you can easily handle complex calculations. It meets standards for #21CFRPart11 compliance. ms.spr.ly/6017grk3h
Speed + compliance in one click. SutiSign makes 21 CFR Part 11 eSignatures simple. 🔗 Blog: sutisoft.com/blog/sutisign-… #eSignature #21CFRPart11 #SutiSign
Regulatory Compliance in Clinical Data Management: Useful Tips for Success Click the link below to read more linkedin.com/feed/update/ur… #Genbioca #21CFRPart11 #ICHE6R2 #CDM #TMF #CRA #GCP #DMP #EDC

Make 21 CFR Part 11 compliance a breeze with Cyret. Enjoy the benefits of secure audit trails, digital signatures, and a paperless approach, all powered by Oracle ERP. Get in touch with Cyret today Contact: [email protected] Learn more: cyret.com #21CFRPart11

Read our latest blog post to learn why a digital signature solution like #ZohoSign would complement the healthcare industry. 😀 🔖 zurl.co/IOva #DigitalSignature #21CFRPart11

Ensuring compliance & data integrity is critical in pharma A 21 CFR Part 11 compliant DMS enables secure document management with audit trails, e-signatures, and full traceability,simplifying audits and improving efficiency. 👉 amplelogic.com/gamp-solutions… #Pharma #DMS #21CFRPart11
amplelogic.com
DMS | AmpleLogic
Document management system
Congrats Nate!! This is just the #21cfrpart11 compliant EDC I've been looking for
Speed + compliance in one click. SutiSign makes 21 CFR Part 11 eSignatures simple. 🔗 Blog: sutisoft.com/blog/sutisign-… #eSignature #21CFRPart11 #SutiSign
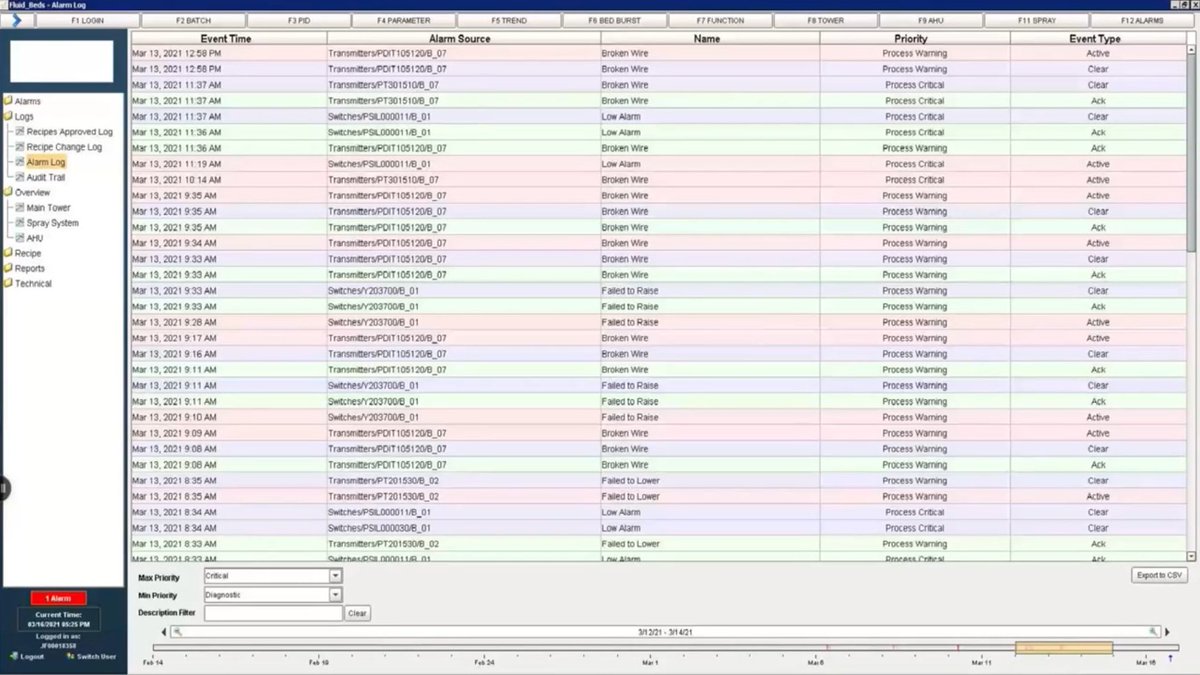
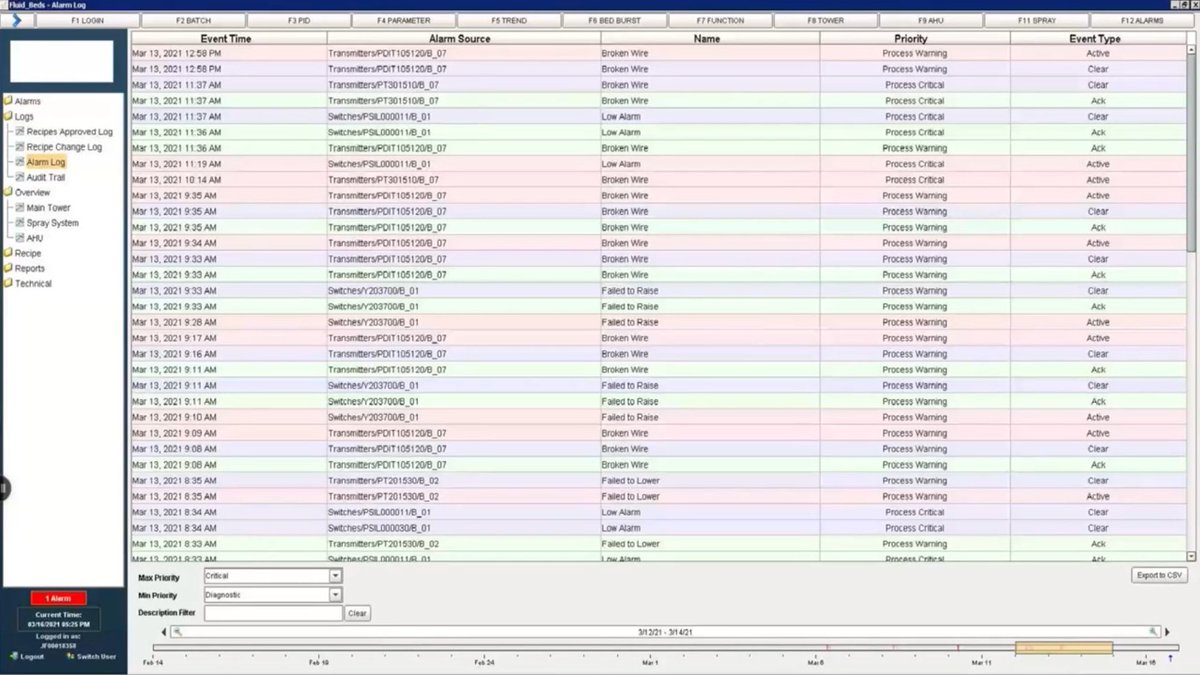
#Pharmaceutical #CaseStudy: Ignition used as a centralized data repository & #21CFRPart11/data integrity #compliance tool PROJECT SCOPE: - 4,367 tags - 49 screens - 92 alarms - Standard architecture - MySQL database See the full case study: bit.ly/48TVJWy
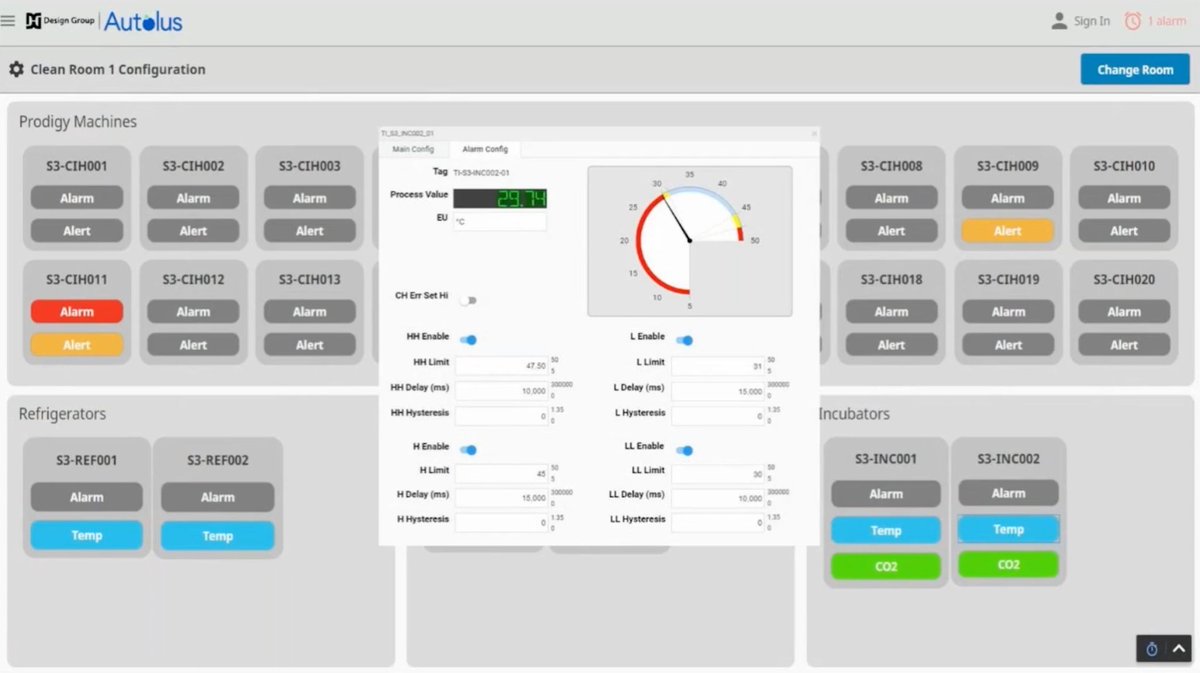
#CaseStudy: @BW_DesignGroup & Autolus Therapeutics: Ignition in the personalized #medicine industry used to handle multiple communication protocols in one #21CFRPart11-compliant environment. Learn more: bit.ly/3twoUyV

📄 Need to meet FDA’s 21 CFR Part 11 for electronic records & signatures? GyrusAim LMS makes it simple with secure e-signatures, audit trails & identity verification. 🔗 Read how: zurl.co/dzAmZ #21CFRPart11 #FDACompliance #LMS #LifeSciences #GyrusAim
gyrus.com
21 CFR Part 11 Compliance for Electronic Records and Signatures
Achieve 21 CFR Part 11 compliance for electronic records and signatures using GyrusAim LMS. Secure e-signatures, audit trails, & authentication.
#Pharmaceutical #CaseStudy: Ignition used as a centralized data repository & #21CFRPart11/data integrity #compliance tool PROJECT SCOPE: - 4,367 tags - 49 screens - 92 alarms - Standard architecture - MySQL database See the full case study: bit.ly/48TVJWy

21 CFR Part 11: more than just compliance, it’s your audit armor. Implement or upgrade your environmental monitoring with Rees EMS to fully align with 21 CFR Part 11 requirements. Read the full blog here: hubs.li/Q03tzj7z0 #rees #ems #21CFRPart11

#CaseStudy: @BW_DesignGroup & Autolus Therapeutics: Ignition in the personalized #medicine industry used to handle multiple communication protocols in one #21CFRPart11-compliant environment. Learn more: bit.ly/3twoUyV

Is your lab 21 CFR Part 11 compliant? One missed detail could cost more than you think. Implement or upgrade your environmental monitoring with Rees EMS to fully align with 21 CFR Part 11 requirements. Read the full blog here: hubs.li/Q03tzgK50 #rees #ems #21CFRPart11

Is your lab audit-ready? 21 CFR Part 11 is more than a checkbox—it’s your foundation for trusted electronic records & signatures. Crucial for pharma, biotech, & diagnostics labs. lablynx.com/resources/arti… #21CFRPart11 #LabCompliance #LIMS #LifeSciences #LabManagement
lablynx.com
Ensure 21 CFR Part 11 Compliance with Lab Software Solutions
Meet FDA regulations with secure laboratory software designed for 21 CFR Part 11 compliance. Ensure data integrity, audit trails, and electronic signatures.
Regulated lab? You need software that understands compliance. Here are 5 must-have capabilities for targeted MS quantitation: 🔗 spr.ly/60124ywoA #CDS #MassSpec #21CFRPart11

#CaseStudy: @BW_DesignGroup & Autolus Therapeutics: Ignition in the personalized #medicine industry used to handle multiple communication protocols in one #21CFRPart11-compliant environment. Learn more: bit.ly/3twoUyV
New Blog: 21 CFR Part 11 in Life Sciences: Core Requirements & Industry Best Practices From e-signatures to audit trails, get expert insights on staying compliant in the digital age of #LifeSciences. Read now: hubs.li/Q03s1w7m0 #21CFRPart11 #Compliance #FDA #Rees
#Pharmaceutical #CaseStudy: Ignition used as a centralized data repository & #21CFRPart11/data integrity #compliance tool PROJECT SCOPE: - 4,367 tags - 49 screens - 92 alarms - Standard architecture - MySQL database See the full case study: bit.ly/48TVJWy

Is your LMS helping you stay 21 CFR Part 11 compliant? 📽️ Watch our quick explainer on how life sciences teams can streamline compliance training with the right LMS: zurl.co/V7Uo6 #LifeSciences #21CFRPart11 #ComplianceTraining #LMS #eLearning
youtube.com
YouTube
21 CFR Part 11 LMS Compliance in Life Sciences
Did you know? Many 21 CFR Part 11 violations stem from weak access controls and poor audit trails. To reduce your risk: •Enforce least privilege •Implement strong authentication •Monitor and log access regularly #CyberSecurity #LifeSciences #21CFRPart11 #HIPAA
Validation isn’t red tape—it’s risk management. The stronger your validation, the more confident your lab becomes. How strong is your current CSV approach? #CSV #21CFRPart11 #ComplianceMatters
#Pharmaceutical #CaseStudy: Ignition used as a centralized data repository & #21CFRPart11/data integrity #compliance tool PROJECT SCOPE: - 4,367 tags - 49 screens - 92 alarms - Standard architecture - MySQL database See the full case study: bit.ly/48TVJWy
Webinar Insights "21 CFR Part 11 Compliance with Electronic Signatures" youtube.com/watch?v=fsopE4… Register to watch the full video conferencepanel.com/conference/21-… #conferencepanel #21cfr #21cfrpart11 #electronicsignature #softwareasaservice #annex11 #electronicrecords #dataintegrity #gmp

youtube.com
YouTube
21 CFR Part 11 Compliance with Electronic Signatures - 2025 Updates
Our lab solutions are like having ten smart lab assistants, except they never take a day off. #labautomation #dataintegrity #21cfrpart11 #laboratory #software

📣 We are live! Join us now in our live showcase to learn how to ensure #DataIntegrity of Printed Documents with Scilife. Access directly here ➡️ bit.ly/prshowcase #21CFRPart11 #Annex11

#Celltherapy manufacturing & #21CFRPart11 compliance: Overview & considerations for instrument software In this #webinar, @thermofisher will share insights to help you better understand how electronic records & 21 CFR Part 11 compliance are related bit.ly/3lzrH6y

qmsWrapper, Quality Management Software, fully supports 21 CFR Part 11. With electronic signatures, you can trust the authenticity and integrity of your records. Elevate your compliance game with qmsWrapper. Learn more today! buff.ly/2lmOBhW #QMS #Compliance #21CFRPart11

Regulatory Compliance in Clinical Data Management: Useful Tips for Success Click the link below to read more linkedin.com/feed/update/ur… #Genbioca #21CFRPart11 #ICHE6R2 #CDM #TMF #CRA #GCP #DMP #EDC

21 CFR Part 11: more than just compliance, it’s your audit armor. Implement or upgrade your environmental monitoring with Rees EMS to fully align with 21 CFR Part 11 requirements. Read the full blog here: hubs.li/Q03tzj7z0 #rees #ems #21CFRPart11

Say goodbye to outdated testing methods! Upgrade to the 21 CFR Part 11 Compliant DVNext Full Feature Rheometer and revolutionise your viscosity and yield stress testing. #Rheometer #TestingUpgrade #21CFRPart11 okt.to/ayPJsB?

The Pharma Training Hyderabad thepharmatraininghyderabad.com #21CFRPart11 #CSVPharma #CSVGlobalJobs #CSVAbroadCareers #DataIntegrityCompliance #PharmaComplianceJobs #BestCSVInstituteHyderabad #PharmaValidation #CSVExpertTraining #PharmaITCareers #PharmaIndustryJobs #PharmaJobsAbroad

Is your biobank compliant with key regulations and standards like #GDPR, #HIPAA, #21CFRPart11, & ISO 20387? Discover how #LIMS simplifies compliance and enhances secure data management. Stay ahead with streamlined processes and full regulatory adherence. cloudlims.com/gdpr-hippa-21-…

Enhance your understanding of 21 CFR Part 11 compliance with our course on The GAMP Approach to 21 CFR Part 11 Compliance. Course Highlights: Key aspects of 21 CFR Part 11 Applying the GAM... eleap.me/4esWRD8 #eLeaP #ComplianceTraining #21CFRPart11 #GAMPApproach #Elearning

Read our latest blog post to learn why a digital signature solution like #ZohoSign would complement the healthcare industry. 😀 🔖 zurl.co/IOva #DigitalSignature #21CFRPart11

This webinar with David Nettleton, Learn how to use #microsoftexcel spreadsheets for #GXP data to ensure compliance with #21cfrpart11 and reduce #validation cost and time. bit.ly/3ljtCMb

Ensuring Regulatory Compliance: #Software Solutions for Adhering to #MedicalStandards. #21CFRPart11 #FDA #ETMF #Solutions Read about it here: bluecord.com/post/ensuring-…

New case study! 🚀 See how we transformed a legacy instrument software into a 21 CFR Part 11-ready solution with secure user management, audit trails & e-signatures, without hurting usability. hubs.ly/Q03TYWSh0 #BioSistemika #LabSoftware #21CFRPart11

Is your lab 21 CFR Part 11 compliant? One missed detail could cost more than you think. Implement or upgrade your environmental monitoring with Rees EMS to fully align with 21 CFR Part 11 requirements. Read the full blog here: hubs.li/Q03tzgK50 #rees #ems #21CFRPart11

The Power Of A No Code Platform & Solutions, It Really Matters! cloudtheapp.com/power-of-a-no-… #21CFRPart11 #Cloudtheapp #Compliance #Configurable #NoCode #QMS #QualityManagementSystem #Validation

Pharma signing demands intent, accountability, and full traceability—not just a digital signature. CryptoESIGN CFR ensures every approval is compliant, auditable, and regulation-ready from start to finish. cryptoesign.com #CryptoSIGN #21CFRPart11 #digitalSignatures

Get the key aspects of #21cfrpart11 validation and maintaining covered. Join the webinar by best in the industry Carolyn Troiano and prepare your team to meet both #fda and International compliance requirements. lnkd.in/gekQ7zem #webinar #gdpr #compliancetrainings

The Bedrock of Trust: GenBioCa's Commitment to Global Regulatory Compliance! linkedin.com/feed/update/ur… Visit genbioca.com or mail us at [email protected] #RegulatoryCompliance #FDA #21CFRPart11 #DataIntegrity #ClinicalTrials #QualityAssurance #GenBioCa

Make 21 CFR Part 11 compliance a breeze with Cyret. Enjoy the benefits of secure audit trails, digital signatures, and a paperless approach, all powered by Oracle ERP. Get in touch with Cyret today Contact: [email protected] Learn more: cyret.com #21CFRPart11

Something went wrong.
Something went wrong.
United States Trends
- 1. Arsenal N/A
- 2. $RISE N/A
- 3. Arteta N/A
- 4. Jayson Tatum N/A
- 5. Sixers N/A
- 6. Haaland N/A
- 7. Gabriel N/A
- 8. Shreveport N/A
- 9. Havertz N/A
- 10. Maxey N/A
- 11. #MCIARS N/A
- 12. #HBDRizzlerGiveaway N/A
- 13. Cherki N/A
- 14. Man City N/A
- 15. Odegaard N/A
- 16. #DifferentHere N/A
- 17. #Celtics N/A
- 18. #JAYKeepsTheENGENEsON N/A
- 19. Donnarumma N/A
- 20. Bernardo Silva N/A


































