#fdafasttrack search results
Priovant Therapeutics has begun a phase 3 trial evaluating brepocitinib as a uveitis treatment. Read more: ow.ly/IHeG50TCnrc #uveitis #retinalphysician #FDAfasttrack

#regulatory #news ~@genprex receives #FDAFastTrack for REQORSA® #immunogene rtherapy ~@BioMarin wins #FDAApproval for first #HemophiliaA #GeneTherapy ~FDA Approves @ARUPLabs' AAV5 DetectCDx™, first-companion diagnostic #Immunoassay for #GeneTherapy

🚨 Final Session Alert! 🚨 Join us Tue, July 15 | 12–1 PM (PDT) on Zoom for FDA Programs to Expedite Drug Development with Elizabeth Gordon, sponsored by RAIN Catalysts. Free to attend. Register now: zurl.co/nR7ku #FDAFastTrack #DrugDevelopment #BiotechEvents

Pulmonary NTM infection is recognized as a major global health concern due to its rising prevalence worldwide, association with shortened life span and significant impact on patients’ daily living. #MannKind #FDAFastTrack #NTMLungDisease Learn more at bit.ly/3wpEGxh

FDA fast-track = speed. But success = data. Here are 6 life science workforce metrics that help you hire faster, stay compliant, and scale smart. 📊hubs.li/Q03ykYrw0 #LifeSciences #FDAFastTrack #WorkforceAnalytics #SOW #ICCompliance #SunaSolutions
suna.com
Life Science Workforce Metrics to Follow After FDA Fast Tracking - Suna
Life science workforce metrics every leader should know in regards to FDA fast tracking and how to track 6 integral KPIs.
Shanton Receives Fast Track Designation from US FDA for Refractory Gout Program thingsofbusiness.com/news/065aba1b7… #ShantonPharma #FDAFastTrack #GoutTreatment #RefractoryGout #SAP001 #PharmaNews #BiotechNews #DrugDevelopment #MedicalInnovation #Pharmaceuticals #HealthcareNews
thingsofbusiness.com
Shanton Receives Fast Track Designation from US FDA for Refractory Gout Program
SAP-001 is Shanton's lead investigational compound with First- and Best-in-Class potential in uncontrolled gout SINGAPORE and PRINCETON, N.J., July 25, 2025 -- Shanton Pharma, a clinical-stage...
#regulatory #news ~@AvengeBio receives #FDAFastTrack for #CellTherapy leveraging its #Immunotherapy platform @Calliditas1 files with @MHRAgovuk for Kinpeygo in IgA #nephropathy ~@AlzamendNeuro receives FDA “Study May Proceed” notification for study in #BipolarDisorder

The FDA, under Commissioner Martin Makary, is piloting a voucher program that could expedite drug reviews to one or two months. This fast-track approval process is contingent on the drug meeting specific requirements. biopharmadive.com/news/FDA-natio… #fdaapproval #fdafasttrack #fda
#regulatory roundup ~@Diamyd receives #FDAFastTrack designation for Diamyd® ~FDA approves @SamsungUS’s #sleepapnea feature on #Galaxywatch, phone ~FDA grants clearance to @nanox_vision #AI #software to assess early signs of #fattyliver

We’re excited to share that the FDA has granted Fast Track designation to NS-229 for Eosinophilic Granulomatosis with Polyangiitis (#EGPA), a rare autoimmune disease! Read the full announcement: ow.ly/h95w50WTUFl #RareDiseases #FDAFastTrack #PharmaInnovation

Artiva Biotherapeutics' lupus treatment receives FDA Fast Track designation noah-news.com/gb/en/health/2… #lupusnephritis #FDAFastTrack #immunotherapy
Telix Pharmaceuticals has announced a significant milestone in its development of a new glioma imaging product, TLX101-CDx. #TelixPharmaceuticals #FDAFastTrack #GliomaImaging #TLX101CDx #MedicalInnovation #CancerDiagnosis #BrainTumor #HealthcareNews zurl.co/OhsD
Johnson & Johnson's posdinemab, a tau-targeted monoclonal antibody for early Alzheimer's disease, receives FDA Fast Track. #AlzheimersResearch #JNJ #FDAFastTrack linkedin.com/posts/jnjinnov…
linkedin.com
We’re proud to share that our investigational tau-directed monoclonal antibody (mAb) for the...
We’re proud to share that our investigational tau-directed monoclonal antibody (mAb) for the treatment of early Alzheimer's disease (AD) has received U.S. FDA Fast Track designation. This marks the...
FDA grants Fast Track designation to MAIA’s ateganosine for #NSCLC. The telomere-targeting therapy showed partial responses in phase 2 THIO-101 trial with cemiplimab in patients with advanced NSCLC. Read more via @CancerTherAdvsr: bit.ly/4oMz1HY #Oncology #FDAFastTrack
FDA fast-tracks Empaveli for kidney disease, accelerating its development to address unmet patient needs in renal care. @ApellisPharma 📖 Read more: pharmanow.live/latest-news/fd… #Empaveli #FDAFastTrack #KidneyDisease #RenalCare #PharmaNews #HealthcareInnovation
📢 FDA Fast Tracks TME Pharma's Glioblastoma Treatment #TMEPharma #FDAFastTrack #Glioblastoma #NOXA12 bioprocessupdates.com/companies/fda-…
Ensysce has earned both “Fast Track” and “Breakthrough Therapy Designation” (BTD) from the FDA! What do they mean? Check out this piece on the #FDA site about BTD. #fdafasttrack #fdabtd #bioscience #opioidpharma fda.gov/regulatory-inf…
Did you know that Ensysce has earned “Fast Track” designation from the FDA? What does that mean? Check out this piece on the #FDA site. fda.gov/patients/fast-… #fdafasttrack #bioscience
@CalidiBio CLD‑201 earns @US_FDA Fast Track designation for soft tissue sarcoma. A key step forward for their stem cell–based virotherapy platform. 🔗tipranks.com/news/the-fly/c… #Biotech #FDAFastTrack #Oncology #Immunotherapy #CancerResearch
Incannex Healthcare secures FDA Fast Track designation for IHL-42X in obstructive sleep apnea treatment market business-news-today.com/incannex-healt… #IncannexHealthcare #IHL42X #FDAFastTrack #SleepApnea #BiotechStocks #ClinicalTrials #HealthcareInnovation
Sangamo Therapeutics wins FDA fast-track status for gene therapy ST-503 targeting small fiber neuropathy business-news-today.com/sangamo-therap… #SangamoTherapeutics #FDAFastTrack #GeneTherapy #SmallFiberNeuropathy #Biotechnology #Neurology #ChronicPain
business-news-today.com
Sangamo Therapeutics wins FDA fast-track status for gene therapy ST-503
Find out how Sangamo Therapeutics’ FDA fast-track win for ST-503 could reshape small fiber neuropathy treatment and shift biotech investor sentiment.
Neuren Pharmaceuticals secures third FDA Fast Track for NNZ-2591 as shares rise 4.21% business-news-today.com/neuren-pharmac… #NeurenPharma #NNZ2591 #FDAFastTrack #NeurenPharmaceuticals #OrphanDrugs #RareDiseases #ASXBiotech #PMSAwareness #PediatricNeurology #KoalaTrial #AcadiaPharma
Check out this article from Rare Cancer News on CER-1236, #CeroTherapeutics’ lead therapy for #AML, receiving #FDAFastTrack designation. CER-1236 uses CER T-cells to kill #cancercells & enhance the immune response. Full article: bit.ly/4pmOJtF $CERO #ClinicalTrials
We’re excited to share that the FDA has granted Fast Track designation to NS-229 for Eosinophilic Granulomatosis with Polyangiitis (#EGPA), a rare autoimmune disease! Read the full announcement: ow.ly/h95w50WTUFl #RareDiseases #FDAFastTrack #PharmaInnovation

FDA grants Fast Track designation to MAIA’s ateganosine for #NSCLC. The telomere-targeting therapy showed partial responses in phase 2 THIO-101 trial with cemiplimab in patients with advanced NSCLC. Read more via @CancerTherAdvsr: bit.ly/4oMz1HY #Oncology #FDAFastTrack
@CalidiBio CLD‑201 earns @US_FDA Fast Track designation for soft tissue sarcoma. A key step forward for their stem cell–based virotherapy platform. 🔗tipranks.com/news/the-fly/c… #Biotech #FDAFastTrack #Oncology #Immunotherapy #CancerResearch
Shanton Receives Fast Track Designation from US FDA for Refractory Gout Program thingsofbusiness.com/news/065aba1b7… #ShantonPharma #FDAFastTrack #GoutTreatment #RefractoryGout #SAP001 #PharmaNews #BiotechNews #DrugDevelopment #MedicalInnovation #Pharmaceuticals #HealthcareNews
thingsofbusiness.com
Shanton Receives Fast Track Designation from US FDA for Refractory Gout Program
SAP-001 is Shanton's lead investigational compound with First- and Best-in-Class potential in uncontrolled gout SINGAPORE and PRINCETON, N.J., July 25, 2025 -- Shanton Pharma, a clinical-stage...
FDA fast-track = speed. But success = data. Here are 6 life science workforce metrics that help you hire faster, stay compliant, and scale smart. 📊hubs.li/Q03ykYrw0 #LifeSciences #FDAFastTrack #WorkforceAnalytics #SOW #ICCompliance #SunaSolutions
suna.com
Life Science Workforce Metrics to Follow After FDA Fast Tracking - Suna
Life science workforce metrics every leader should know in regards to FDA fast tracking and how to track 6 integral KPIs.
What does it mean-Ensysce’s PF614 with TAAPTM technology has earned FDA's “Fast Track” designation? It's designed to accelerate the process toward commercialization of select breakthrough therapies. en.wikipedia.org/wiki/Fast_trac… #fdafasttrack #bioscience #opioidpharma #pharmafasttrack

Did you know that Ensysce’s PF614 with TAAP technology has earned “Fast Track” designation from the FDA? What does that mean? Check out this piece on the #FDA site about the process. fda.gov/patients/fast-… #fdafasttrack #bioscience #opioidpharma

Ensysce in the NEWS: the latest release about the initiation of a 2nd human abuse potential study on PF614 vs immediate release oxycodone & relative potential for drug liking at equivalent drug dosages. ir.ensysce.com/news/press-rel… #fdafasttrack #biosciences #stopprescriptionabuse

UPDATE: encouraging feedback & guidance from FDA for the Clinical Development Pathway for PF614, suggesting Ensysce’s approach appears reasonable to support a new drug application for an acute pain indication ir.ensysce.com/news/press-rel… #fdaguidance #fdafasttrack #clinicaltrials

What does it mean that Ensysce’s PF614 with TAAP technology has earned “Fast Track” designation? Here’s how Wikipedia described the FDA’s “Fast Track” - accelerating the process of select therapies. en.wikipedia.org/wiki/Fast_trac… #fdafasttrack #bioscience #opioidpharma #pharmafasttrack

Status update: the head of the Food and Drug Administration says a government review of prescription opioid medications, including their risks & contribution to the U.S. overdose epidemic, is still underway. usnews.com/news/health-ne… #nextgenpharma #fdafasttrack #prescriptionabuse

The FDA’s “Fast Track” program accelerates the process toward commercialization of select therapies. Did you know that Ensysce’s PF614 with TAAP TM technology has earned “Fast Track” designation? fda.gov/patients/fast-… #fdafasttrack #bioscience #opioidpharma #pharmafasttrack

Pulmonary NTM infection is recognized as a major global health concern due to its rising prevalence worldwide, association with shortened life span and significant impact on patients’ daily living. #MannKind #FDAFastTrack #NTMLungDisease Learn more at bit.ly/3wpEGxh

#regulatory roundup ~@Diamyd receives #FDAFastTrack designation for Diamyd® ~FDA approves @SamsungUS’s #sleepapnea feature on #Galaxywatch, phone ~FDA grants clearance to @nanox_vision #AI #software to assess early signs of #fattyliver

Did you know that Ensysce’s science has earned “Fast Track” designation from the FDA? What does that mean? Check out this piece on the #FDA site about the process. fda.gov/patients/fast-… #fdafasttrack #bioscience #opioidpharma

@FDACommissioner @POTUS @realDonaldTrump ALS Therapies need approval ASAP! #Nurown#Tregs#CuAtsm #FDAapproval #FDAfasttrack #Executiveorder We don't have years to wait!!

@SteveHahnMD @FDACommissioner @US_FDA @POTUS @FLOTUS @JujuChangABC Every year my siblings sit in the same spots on a sofa and take a picture. I want to continue to be in the picture too !! #Nurown #FDAApproval #FDAFasttrack #EndALS #Hearourplea #alstherapy

@SteveFDA @US_FDA @realDonaldTrump @als_now @AmandaS49737509 @stevens_nation @iamalsorg @JujuChangABC ALS Patients everywhere......Running on Empty ! We Need NurOwn in our Bodies !!! #NurOwn #FDAApproval #FDAFasttrack #EndALS

more #regulatory wins ~@SangamoTx receives #FDAFastTrack designation for treatment of #FabryDisease ~@boehringerus Ingelheim receives #FDA approval for Cyltezo ® Pen ~@Hologic wins FDA clearance for #COVID-#flu-#RSV assay

#regulatory #news ~@AvengeBio receives #FDAFastTrack for #CellTherapy leveraging its #Immunotherapy platform @Calliditas1 files with @MHRAgovuk for Kinpeygo in IgA #nephropathy ~@AlzamendNeuro receives FDA “Study May Proceed” notification for study in #BipolarDisorder

@FDACommissioner @US_FDA @SteveHahnMD @realDonaldTrump @HHSGov @USCongress Hurry up and Save Lives ! NUROWN APPROVAL #NUROWN #FDAFASTTRACK #FDAAPPROVAL #ENDALS
#regulatory wins ~@Dyne_tx receives #FDAFastTrack for #DuchenneMuscularDystrophy treatment ~@antengene receives #FDA #IND clearance for #SmallMolecule trial ~@AlzamendNeuro receives FDA “Study May Proceed” letter for #Immunotherapy #vaccine for mild-moderate #Dementia

@SteveFDA @realDonaldTrump @als_now @stevens_nation Pandemic.....Yes.......BUT...... LISTEN ALS loses 6000 lives EVERY Year !! 30,000 ALS Patients currently dying ALS kills an American every 90 min. Wake Up FDA !! We Matter Too !! #Nurown #FDAApproval #FDAFasttrack
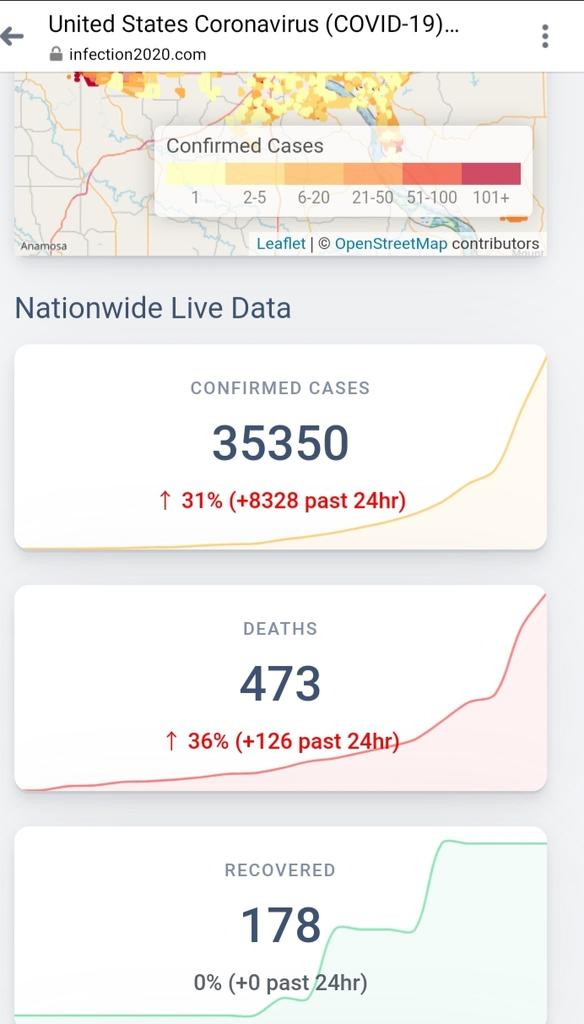
Something went wrong.
Something went wrong.
United States Trends
- 1. Azzi N/A
- 2. Elvis N/A
- 3. UConn N/A
- 4. Syracuse N/A
- 5. Graceland N/A
- 6. Election Day N/A
- 7. Hannah Hidalgo N/A
- 8. #ENHYPEN_ALWAYS_7 N/A
- 9. Star Trek N/A
- 10. Solange N/A
- 11. The ACA N/A
- 12. #HEESEUNG_Belongs_With_ENHYPEN N/A
- 13. Randy Bennett N/A
- 14. Moana N/A
- 15. Obamacare N/A
- 16. Wonder Man N/A
- 17. Starfleet Academy N/A
- 18. Homeward Bound N/A
- 19. Tommie N/A
- 20. Elmo N/A






























