#cdrh search results
⚠️ Medical Devices News: The (#FDA) Center for Devices and Radiological Health (#CDRH) will be transitioning from paper export documents for #medical devices to electronic export documents starting January 2, 2024. For more information ⏩ bit.ly/46WXRf5

📢MEDICAL DEVICES NEWS El Centro de Dispositivos y Salud Radiológica (CDRH) de la FDA publicó su Informe Anual 2023, que destaca los logros alcanzados. 🔗 Lee el reporte completo aquí: bit.ly/49umSza #MedicalDevices #Regulation #CDRH #FDA #DispositivosMédicos

#FDA #CDRH official: Novel #medicaldevices submissions show #medtech sector strength post-COVID | #regulatory raps.org/news-and-artic…
raps.org
CDRH official: Novel device submissions show medtech sector strength post-COVID | RAPS
Now that the nation has transitioned out of the emergency phase of the COVID-19 pandemic, top officials at the US Food and Drug Administration@s (FDA) device center say they have seen a @resurgence@...
Tarver asks for patience from industry as #CDRH undergoes changes | #FDA #healthcare #regulatory #medicaldevices buff.ly/sBa1uNf
#FDA #CDRH shares #regulatory guidance priorities for the coming 12 mo - Use of real-world evidence #RWE to support regulatory decisions and predetermined change control plans for medical devices are two areas of focus for the agency medtechdive.com/news/fda-regul…
medtechdive.com
CDRH shares regulatory guidance priorities for the coming 12 months
Use of real-world evidence to support regulatory decisions and predetermined change control plans for medical devices are two areas of focus for the agency.
#CDRH on track for review timelines despite staff cuts | #FDA #medicaldevices #regulatory buff.ly/s1EmUNo
#FDA #CDRH targets hospital #readmissions with #homehealth #medicaldevice #innovation challenge - The center plans to pick nine devices to help support patients and caregivers after a hospital stay. medtechdive.com/news/cdrh-targ…
medtechdive.com
CDRH targets hospital readmissions with home device innovation challenge
The center plans to pick nine devices to help support patients and caregivers after a hospital stay.
Current #NHLBI awardees that are developing a technology under the regulatory authority of the #CDRH and have not met with FDA previously are eligible to participate. 3/4
Unpacking Averages: #CDRH Recognition of Consensus Standards Appears to Overlook Software #AI #law bit.ly/3UFYyoz @AshleySalvino

FDA’s Center for Devices and Radiological Health recently released a set of three draft guidance documents to help modernize its 510(k) Premarket Notification program. Our alert takes a closer look. #FDA #CDRH #ClinicalData #ImplantDevices dlapiper.com/en/insights/pu…

#FDARegulatoryNewsandTrends: #CDRH finalizes guidance addressing device remanufacturing and servicing; #FDA reorganization of Human Foods Program and other modernization efforts are approved. #Nutrition #REMS #VeterinaryProducts #Project5In5 #GRAS spr.ly/601258D4Q

#QSMR is here...2Harris can help in all of your compliance needs #FDA #CDRH fda.gov/medical-device…
fda.gov
Quality Management System Regulation: Final Rule - FAQ
Information pertaining to the Final Rule that amended the Quality System regulation that is now the Quality Management System Regulation (QMSR)
The @US_FDA #CDRH Issues 2024 #Safety and #Innovation Reports fda.gov/medical-device…
Have you heard our latest episode on the evolving state of device reviews? Check it out: lnkd.in/gageXTQR Guests Mike Ryan and Michael Gaba dive into the latest developments, including FDA’s elimination of RWE submission requirements—and much more. #FDAWatch #FDA #CDRH
#FDARegulatoryNewsandTrends: Final guidance on #cybersecurity in medical devices; #CDRH releases draft guidance documents for weight loss devices; #BreakthroughDevices Program; and more. #FDA #ConfirmatoryEvidence #PrescriptionDrugs #MoCRA spr.ly/6017uuxG1

Over a year since its initial draft guidance document on the use of PCCPs for medical devices that leverage machine learning, the agency has issued a new draft guidance on the use of PCCPs for all medical devices. Read more: bit.ly/3T12K1h #AgencyIQ #Politico #CDRH

The paper also highlights the need for generalizable and representative #clinicaldata and poses key questions to guide public feedback. The #CDRH is actively seeking public input on these considerations. Submit your comments by October 4, 2024. 3/4 #clinicalstudies
#JobOpening The Center for Devices and Radiological Health #CDRH is #Hiring an Assistant Director (Supervisory Health Communications Specialist)! Learn more and #ApplyNow: fda.gov/media/183387/d… #FDA #FDAJobs #Jobs
Join us for #PegaWorld iNspire sessions to see how the @FDA re-envisioned how #CDRH interacts with medical device manufacturers worldwide, while using a secure cloud-based solution and significantly improving the customer experience. Register Today!... pe.ga/43ETtj1

#FDA #CDRH targets hospital #readmissions with #homehealth #medicaldevice #innovation challenge - The center plans to pick nine devices to help support patients and caregivers after a hospital stay. medtechdive.com/news/cdrh-targ…
This guidance, developed by #CDRH and #CBER, is a key in FDA’s broader commitment to implementing structured, template-driven submission tools that enhance both the consistency and efficiency of the premarket review process. 2/4 #FDAGuidance #QSubmissions #PreSubmissions
Tarver asks for patience from industry as #CDRH undergoes changes | #FDA #healthcare #regulatory #medicaldevices buff.ly/sBa1uNf
The FDA’s Center for Devices and Radiological Health (CDRH) released their 2024 Annual Report, showcasing accomplishments in patient-centered initiatives, device safety, and more. Full report here: ow.ly/S2bU50UKKyh #WeSpeakScience #medicalwriting #CDRH #medicaldevices

El #CDRH de la #FDA implementa un piloto que proporcionará alertas tempranas sobre posibles retiros o correcciones de dispositivos de alto riesgo en áreas clave como #enfermedades cardiovasculares, gastro-renales, obstetricia, ginecología y urología. fda.gov/medical-device…

Unpacking Averages: #CDRH Recognition of Consensus Standards Appears to Overlook Software #AI #law bit.ly/3UFYyoz @AshleySalvino

#JobOpening The Center for Devices and Radiological Health #CDRH is #Hiring an Assistant Director (Supervisory Health Communications Specialist)! Learn more and #ApplyNow: fda.gov/media/183387/d… #FDA #FDAJobs #Jobs
Unpacking Averages: #CDRH Recognition of Consensus Standards Appears to Overlook Software #AI #law bit.ly/3UFYyoz @EpsteinBecker

Join a free Town Hall on Sept 24 to meet #FDA's new Acting #CDRH Director, Dr. Michelle Tarver, MD, PhD, with insights from Dr. Jeff Shuren. Learn about her plans for industry engagement, benefit/risk, and innovation. Register now: lnkd.in/gtEwAuw9 #MedTech
The @US_FDA s #CDRH announced in their final guidance that starting October 2025, #medicaldevice manufacturers will be required to use the electronic Submission Template and Resource #eSTAR for all de novo applications. 1/5 fda.gov/media/172450/d… #MedicalDevices #DeNovo

Over a year since its initial draft guidance document on the use of PCCPs for medical devices that leverage machine learning, the agency has issued a new draft guidance on the use of PCCPs for all medical devices. Read more: bit.ly/3T12K1h #AgencyIQ #Politico #CDRH

The paper also highlights the need for generalizable and representative #clinicaldata and poses key questions to guide public feedback. The #CDRH is actively seeking public input on these considerations. Submit your comments by October 4, 2024. 3/4 #clinicalstudies
The @US_FDA s #CDRH has released a discussion paper titled Health Equity For #MedicalDevices. This initiative aims to drive #innovation that reduces barriers to #healthequity and improves outcomes across diverse populations. 1/4 fda.gov/media/180608/d… #PublicHealth #Innovation

@FDADeviceInfo Director Dr. Jeff Shuren announced his plan to retire from the #FDA. He has transferred to the commissioner’s office and Dr. Michelle Tarver, deputy center director for transformation, will assume the role of #CDRH acting director. ow.ly/eymj50SMK9X


#FDARegulatoryNewsandTrends: #CDRH finalizes guidance addressing device remanufacturing and servicing; #FDA reorganization of Human Foods Program and other modernization efforts are approved. #Nutrition #REMS #VeterinaryProducts #Project5In5 #GRAS spr.ly/601258D4Q

5/ Also, it is crucial to identify the patient data distribution the synthetic data is intended to represent. @US_FDA #regulatoryscience @FDADeviceInfo #osel #cdrh
The @US_FDA has announced the launch of the Home as a Health Care Hub to help #healthcare executives design programs that deliver care in the home setting: healthleadersmedia.com/innovation/und… #homehealthcare #CDRH

EU Eyes Transition, #CDRH Builds a Home, Medicare Picks, and More In this week’s Pathways Picks: EU #MDR group meets in advance of May 26 submission deadline; guidance documents related to #EU performance studies and clinical investigations; and more: bit.ly/3wfrNFJ

The @US_FDA #CDRH Issues 2024 #Safety and #Innovation Reports fda.gov/medical-device…
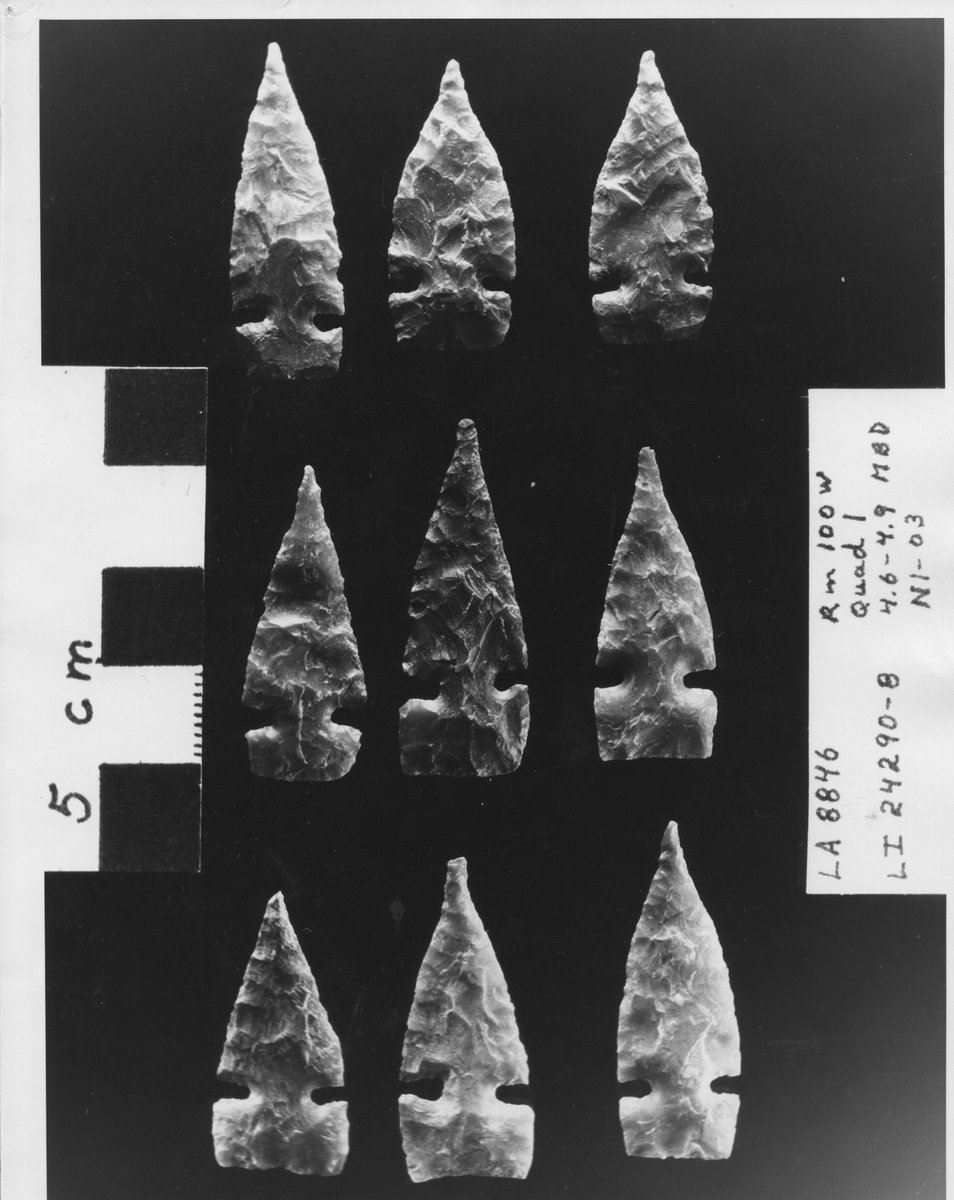
Find pictures of artifacts from the past on the Salmon Pueblo Collection, where thousands of pictures are held for the preservation of a rich culture. salmonpueblo.org #UNL #CDRH

CDRH Review delays are now very visible. Resources shifted to battle COVID mean delays in pre-subs & 510(k) reviews. From IVD groups affected last fall, the delays spread to other groups. graematterintel.com/covid19anddevi… #CDRH #FDA #MedDevice #RegIntel #RegIQ

Plenary session on new directions in DH kicks of inaugural Current Research in Digital History conference, hosted by @chnm #cdrh

Seriously?! Exempt 83 med devices based only on # of MDRs? HHS proposed this in January. Bad analysis approach! Today FDA rightly says "no" - FR notice explains the faulty analysis well. graematter.egnyte.com/dl/1zhdlUlFwo #CDRH #MedDev #RIQ #RegIntel

Unpacking Averages: #CDRH Recognition of Consensus Standards Appears to Overlook Software #AI #law bit.ly/3UFYyoz @AshleySalvino

This week, we are proud to feature one of our own student workers from a CDRH project. Jamison Wyatt is a post graduate student working on the Cather archive. Find out more about him in this thread! #UNL #CDRH

With our current journey in the past, our next stop is the “Journals of the Lewis and Clark Expedition”, a collection spanning over 5000 unique journal pages and more. #UNL #CDRH lewisandclarkjournals.unl.edu

FDA inspection issues?! New pre-sub guidance adds meeting for feedback to help prep response. Able to schedule in time?! Hmmm. Also Safer Tech Program (STeP) meetings added. Copy with highlights posted here: graematter.egnyte.com/dl/f88vuEPUIZ #FDA #CDRH #RegIntel #RIQ

Will CDRH review resources be freed up soon? With so many EUAs, the "no alternatives" criteria may be no longer the case. Good insight by Jeff Shapiro. See the FDA Law Blog post here: fdalawblog.net/2021/02/beware… #FDA #CDRH #RegIntel #RIQ

Struggle understanding FDA requirements? Join FREE FDA Conference! Regulatory education from the regulators for the regulated! July 19-23. Separate tracks for drugs, devices, biologics. Agenda here graematter.egnyte.com/dl/2Vok7CKdW3 #FDA #RegIntel #CDRH #CDER #CBER

The CDRH is happy to welcome the “Ardhi Initiative” as part of this week’s featured project. This new initiative aims to shed light on hundreds of years of land-acquisition in Africa and more. Discover more about this new project here! ardhi.unl.edu #UNL #CDRH

Proposed rule to shift from 820 to 13485 has arrived! Does anyone believe it will be as easy as it looks in this notice? Once final, only one year to implement! Better get started soon. graematter.egnyte.com/dl/CWCzaLLB7e #CDRH #FDA #RegIntel #QMS #QSR

What were the conservation efforts of the past in Nebraska? Find out in the Nebraskaland archive, where many issues tackle how to preserve the beautiful wildlife of our home. nebraskaland.unl.edu #UNL #Nebraskaland #CDRH

Prepare yourself for more Breakthrough Device Designation requests! CMS finalizes rule for accelerated coverage. Faster decisions. Coverage up to 4 years. Nice! Fact sheet & FR Notice here: graematter.egnyte.com/fl/ldrO3rwFjy #FDA #CMS #CDRH #RegIntel

Help students learn about life in the Midwest for Mexican-Americans with Family Letters. With many lesson plans, students can learn both history and Spanish in meaningful ways. familyletters.unl.edu/en #UNL #CDRH

Have a project on the historic duo of Lewis and Clark? Why not use this week’s featured project, the "Journals of the Lewis and Clark Expedition"? Find everything you would need for them here! lewisandclarkjournals.unl.edu #UNL #CDRH
The @US_FDA has announced the launch of the Home as a Health Care Hub to help #healthcare executives design programs that deliver care in the home setting: healthleadersmedia.com/innovation/und… #homehealthcare #CDRH

Hear updates from the #FDA offices including #CDER, #CDRH, Office of New Drugs, Rare Diseases Program and Safety, Efficacy and Post Market Surveillance at #FDACMS, December 11-12,2018 lifesciences.knect365.com/fda-cms/?utm_s…

Exemptions for Unclassified Devices: #CDRH Updates Guidance buff.ly/2RRaj8u vía @RAPSorg #medicaldevices

Something went wrong.
Something went wrong.
United States Trends
- 1. #WrestleMania N/A
- 2. Luke Kennard N/A
- 3. #ImmortalCF N/A
- 4. Bengals N/A
- 5. Lakers N/A
- 6. #LakeShow N/A
- 7. #Toonami N/A
- 8. Rockets N/A
- 9. SWEET SPOT N/A
- 10. Sengun N/A
- 11. Dexter Lawrence N/A
- 12. Giants N/A
- 13. #TokyoRevengers N/A
- 14. Porter Martone N/A
- 15. Orton N/A
- 16. Bianca N/A
- 17. Ayton N/A
- 18. Bron N/A
- 19. Trey N/A
- 20. Paige N/A



































































